The early morning cup of tea starts with a cup of water and a teabag separately. The diffusion process is the reason behind the teacup diffusing in the water and changing its color. We observe numerous instances of diffusion in our daily life. To grasp the concept of diffusion better, along with its types, causes and significance, let’s read on.
What is Diffusion?
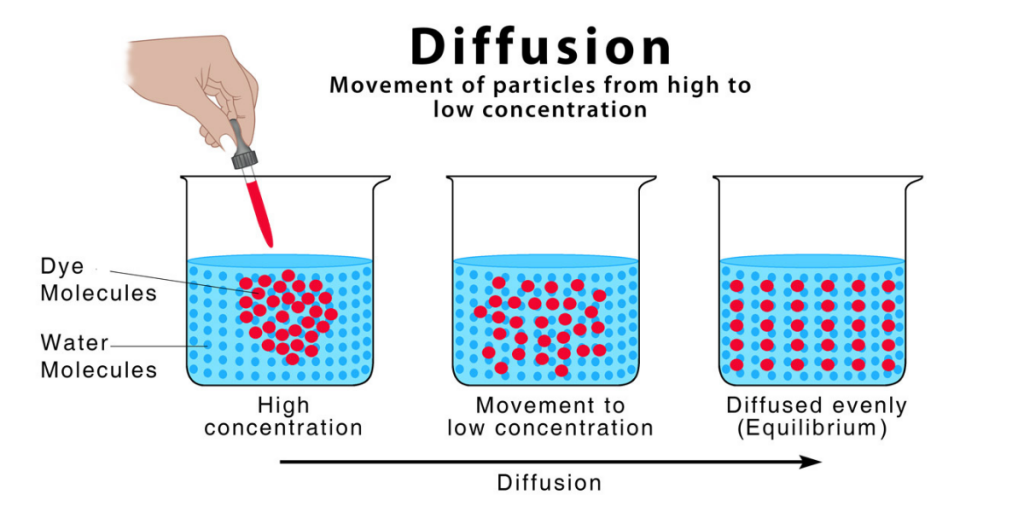
The word Diffusion originates from the Latin word diffusionem and diffusio, which means ‘pouring out’ or ‘pouring forth.’ We observe diffusion in major spheres of science such as physics, chemistry, and biology. Diffusion refers to the passive movement of molecules or particles from a higher contraction region to a lower concentration region.
The process of diffusion occurs everywhere, starting from our internal body parts, such as our kidneys. Diffusion is involved in removing urea and excess fluid from the blood through our kidneys. The concentration gradient of regions is responsible for inciting the molecules to diffuse.
Diffusion Vs. Osmosis: What differentiates diffusion from osmosis, even though they are both types of passive transport, is disseminating particles or molecules. In diffusion, the solutes are the diffusing/disseminating molecules. While in osmosis, the particles are the solvents.
Diffusion Vs. Active Transport: Diffusion is a type of passive transport. In the diffusion process, there is a net movement of molecules downhill along with the steepness of the concentration gradient. What differentiates diffusion from active transport lies in the definition. Active transport signals the movement of molecules against the concentration gradient.
Definition of Diffusion
In biology, the term diffusion is used to describe the physical process of net movement of particles or molecules when they travel from a region of higher concentration to lower concentration. The movement of these molecules in the process of diffusion occurs as a passive transport down the concentration gradient.
Types of Diffusion
The diffusion process is subdivided into two major types or categories. The two major types of diffusion are; simple diffusion and facilitated diffusion. The difference between these types of diffusion lies in their description itself.
- Simple Diffusion: In simple diffusion, molecules transfer through a biological semipermeable membrane or compound without transport membrane proteins playing any part. In other words, transport membrane proteins are not involved in aiding in the process of simple diffusion.
- Facilitated Diffusion: Movement through the semipermeable biological membrane is out of the question for polar and large molecules. Thus, facilitated diffusion requires the aid of plasma membranes. The protein membranes facilitate the transfer of molecules in and out of a cell. Thus, the protein-membrane acts as a carrier molecule in the process of facilitated diffusion.
The major difference between the two types of diffusion stems from the mechanism by which molecules or particles move. The description of the types shows an incisive difference; one takes assistance in the process of diffusion, while the other does not.
To be clear, both types of diffusion eventually lead to a net downhill movement of molecules due to the inclination of the concentration gradient. Moreover, neither of them requires chemical energy to aid in the process of diffusion.
Facilitated diffusion requires the aid of carrier molecules in the mechanism of diffusion. Simple diffusion does not require the aid of transport protein to fulfill the mechanism of diffusion.
Factors Affecting Diffusion
Several factors affect the rate and result of diffusion. A spoonful of sugar dissolves faster in a cup of hot water than it does in room temperature water. The difference in the extent of the breakdown of sugar happens due to one of the factors affecting diffusion, which is temperature.
Following are some of the common factors that affect the rate and extent of diffusion;
- Temperature
- Particular/Molecule Size
- The inclination of Concentration Gradient
- Interaction Area
Shaking or stirring a solution is not involved in the process of diffusion because it is supposed to be natural. In our body, molecules diffuse and move through different metabolic and cellular processes due to the process of diffusion.
Significance of Diffusion
Diffusion plays an important role in science and our life. Instances of diffusion take place across all spheres of life and nature. Let us look at some examples of diffusion to understand its significance on earth;
- Role of Diffusion in Plants: Plant cells help diffuse the water from the soil into different parts of the plant. The process of diffusion in a plant starts from the root hair. Without diffusion, sustenance of plant life is out of the question.
- Role of Diffusion in Respiration: Respiration in living organisms involves breathing in the air in and out. The air contains carbon dioxide that gets diffused out of our bodies. This process of diffusion happens in the cell membrane and blood.
Diffusion plays a significant role in physics and chemistry too. The generation of electric charge and fragrance of a flower that spreads through the still air of a room is possible because of diffusion.

